Clinical Trial Liaisons: The “Missing Link” Between Pharma and Clinical Trial Efficiency
Bread and butter. Hydrogen and oxygen. Manufacturer and CRO. Among pairings that have stood the test of time is that of a pharmaceutical manufacturer and its Contract Research Organization (CRO). CROs are trusted to provide mission-critical services for their pharma clients’ drug development projects because of their deep local regulatory expertise, established protocols, and skilled project management. What’s more, CROs reduce the cost of research that helps businesses and institutions meet the needs of the evolving drug and medical device industry.
Often neglected in this equation are the benefits of investigator-specific relationships and the high levels of ongoing site interaction required to optimize patient enrollment and drive clinical trial efficiency. Put simply, delays in patient enrollment come at a substantial cost:
As part of the movement to help pharma more efficiently achieve clinical trial goals and meet research timelines, Amplity is disrupting the traditional pharma/CRO dynamic with the addition of Clinical Trial Liaisons (CTLs), a role designed to complement the important work of Clinical Research Associates (CRAs).
Clinical Trial Liaisons:
The “Missing Link” Between Pharma and Trial Efficiency
It’s no secret that the more attention a manufacturer devotes to each site, the more likely its investigators are to drive the trial forward with urgency and efficiency.
Adding a CTL to the clinical trial mix allows the CRA to remain focused on clinical protocol and data reporting while the CTL concentrates on opening and maintaining channels of communication with the sites and investigators critical to the success of the trial. So while CTLs and CRAs might have many things in common, their objectives are very different.
As described below, the CTL role includes 3 primary functions.
1. Strengthen Relationships with Clinical Trial Sites
In contrast to the CRA, who visits a site on behalf of multiple trials and/or sponsors, the CTL represents a single sponsor. This is an important differentiator since any given healthcare facility could have multiple CRA-monitored clinical trials taking place in parallel.
This single-trial focus enables CTLs to take on a much more prominent role for the trials they manage. For instance, the CTL can establish trial-specific relationships across a wide network of stakeholders and sites, enabling the appropriate sharing of experiences between sites. CTLs develop a deep familiarity with the trial, inclusion/exclusion criteria, etc., all of which can be shared with each site’s treatment team.
CTLs are focused on building and maintaining strong relationships with site staff by establishing steadfast reliability and trust. The process includes regular interaction with the Principal Investigator as well as other site staff. The CTL engages in peer-to-peer conversations on scientific and clinical issues across the clinical team.
Importantly, CTLs represent a long-term connection between the sponsor and the site. Over time, CTLs become emotionally embedded within a site’s clinical team, instilling and maintaining a level of familiarity and comradery with the site team. This ensures that the CTL’s clinical trial remains a priority at the site. Over time, the relationship between the site staff and the CTL can extend to become a relationship between the site staff and the pharmaceutical sponsor, an important long-term win.
2. Accelerate Patient Enrollment, Thereby Accelerating Data Collection
Compared with a CRA, a CTL expends much more effort – quantitatively and qualitatively – with the site study team. Because CTLs have a deep understanding of inclusion/exclusion and other study criteria, they can help the investigator screen patients with greater accuracy and speed. The high-quality scientific exchange delivered by CTLs gives investigators confidence regarding patient selection and management. In the end, this more informed and efficient site recruitment leads to faster data collection.
3. Collect and Disseminate Actionable Insights
Most written CRA reports flow through a long chain of communication before they reach the key sponsor stakeholders, thus delaying information and action. But because CTLs have frequent peer-to-peer exchange and in-depth conversations with staff from multiple sites, the insights they collect tend to be highly strategic. CTLs routinely uncover important observations – about safety or adverse events, for example – that are reported more directly and much faster than written reports completed by CRAs. These actionable insights are crucial sources of intelligence for headquarters staff.
So how does the CTL role play out in real life? The following case study demonstrates the impact possible.
Amplity’s Clinical Trial Liaison Team Exceeds Global Trial Enrollment Target and Timeline by 6 Weeks
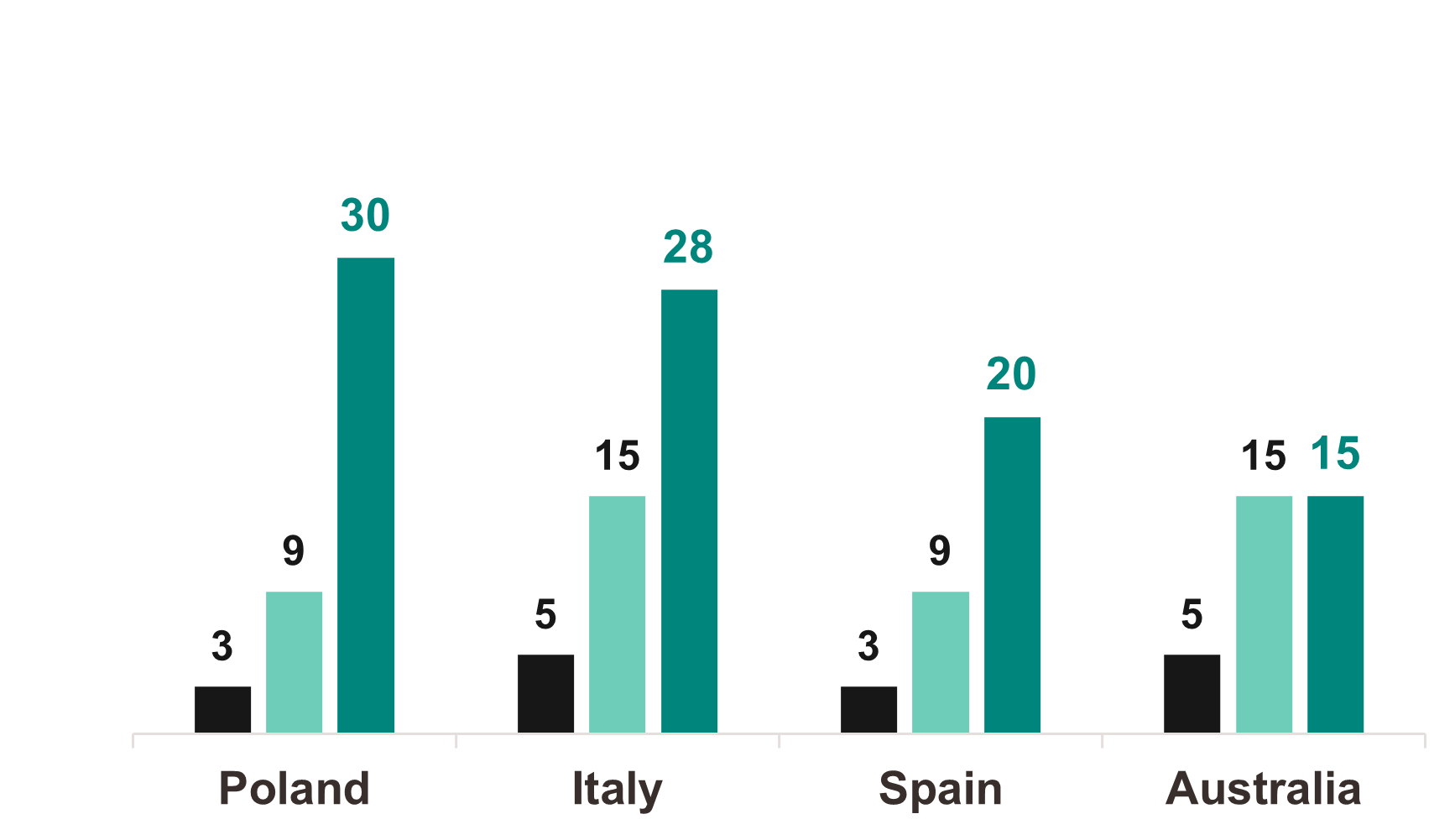
Amplity’s CTLs successfully built site relationships in line with their goals to raise awareness of the trial, the company, and the therapy area, ultimately educating 150+ investigators and their teams. CTLs recruited 221 patients across EU, APAC, and LATAM effectively reaching the global target enrollment goal and exceeding target enrollment timeline by 6 weeks.
Number of desired sites Patient enrollment target Actual patients enrolled
VIEW CASE STUDY
Summary
We all share a key clinical goal as we seek to benefit patients: efficiency. Every day biopharma manufacturers choose to ignore the CRO patient-enrollment speed gap there is a significant cost to the company’s bottom line. While CROs remain important and valuable partners to pharma, CTLs allow CRAs to do what they do best, all while providing indisputable cost-saving potential both in money and time. The Amplity team has been providing CTLs globally for over a decade, and we continue to refine the role to meet the needs of today’s clients.
Ready to discuss how we can help you more efficiently achieve clinical trial goals and meet research timelines?